How Clinical Trials Are Conducted: A Step-by-Step Guide
Discover how clinical trials are conducted step by step with expert insights from clinical research organizations. Learn about the phases, regulatory processes, and trial management with Clinaxis Research.
K. A. Babu
2/18/20263 min read

How Clinical Trials Are Conducted: A Step-by-Step Guide by Clinical Research Organizations (CROs)
Bringing a new drug, medical device, or therapy to market is a long, highly regulated journey. At ClinAxis Research, we partner with sponsors, biotech companies, and pharmaceutical organizations to manage every stage of the clinical trial lifecycle with precision, compliance, and efficiency.
This comprehensive guide explains how clinical trials are conducted step by step, and the critical role Clinical Research Organizations (CROs) play in delivering successful outcomes.
What Are Clinical Trials?
Clinical trials are structured research studies conducted in human participants to evaluate the safety, efficacy, and optimal use of investigational drugs, biologics, or medical devices. These studies follow strict regulatory and ethical guidelines to ensure participant protection and data integrity.
Clinical trials are essential for:
Developing innovative therapies
Ensuring patient safety
Gaining regulatory approval
Advancing medical science
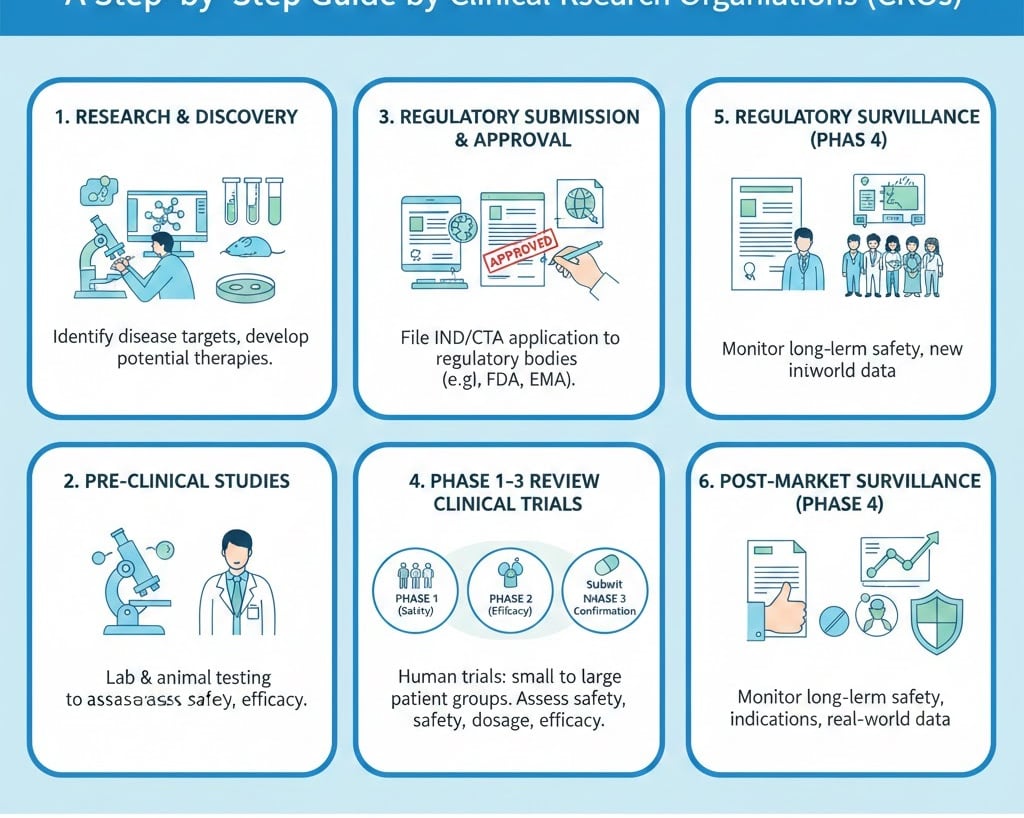
Step-by-Step Process of Conducting Clinical TrialsStep 1: Study Concept and Protocol Development
Every successful clinical trial begins with a strong foundation.
Key Activities:
Identifying research objectives
Defining target patient population
Determining study endpoints
Designing methodology
Risk assessment and feasibility analysis
The study protocol is the blueprint of the trial. It outlines:
Inclusion and exclusion criteria
Study procedures
Dosage and treatment plans
Statistical analysis approach
Safety monitoring plans
At ClinAxis Research, we ensure protocols are scientifically robust, operationally feasible, and aligned with global regulatory standards.
Step 2: Regulatory and Ethics ApprovalsBefore enrolling participants, regulatory clearance is mandatory.
Approval Requirements:
Submission to regulatory authorities (e.g., FDA, EMA, MHRA, CDSCO depending on region)
Institutional Review Board (IRB) / Ethics Committee (EC) approval
Investigational New Drug (IND) or Clinical Trial Application (CTA) submission
CROs manage:
Regulatory documentation
Authority communication
Compliance with ICH-GCP guidelines
Timeline tracking
Our regulatory experts streamline submissions to minimize delays and ensure full compliance.
Step 3: Site Selection and Feasibility
Choosing the right clinical sites significantly impacts trial success.
CRO Responsibilities:
Site identification and qualification
Investigator evaluation
Feasibility assessments
Contract negotiation
Budget management
We evaluate:
Patient recruitment potential
Investigator experience
Infrastructure and resources
Past performance metrics
Strategic site selection improves enrollment speed and data quality.
Step 4: Study Start-Up
This phase prepares sites for patient enrollment.
Activities Include:
Site initiation visits (SIV)
Investigator training
Study material distribution
Electronic Data Capture (EDC) setup
Clinical Trial Management System (CTMS) configuration
Efficient study start-up reduces costly delays and ensures readiness for enrollment.
Step 5: Patient Recruitment and Enrollment
Recruitment is one of the most challenging aspects of clinical trials.
CRO Strategies:
Patient outreach planning
Digital recruitment campaigns
Referral networks
Pre-screening coordination
Retention strategies
We focus on:
Ethical recruitment practices
Clear informed consent process
Diversity and inclusion
Patient-centric strategies improve enrollment timelines and retention rates.
Step 6: Trial Monitoring and Data Management
Once the trial is active, continuous oversight ensures quality and compliance.
Monitoring Includes:
On-site and remote monitoring visits
Source data verification (SDV)
Safety reporting
Protocol compliance checks
Risk-based monitoring approaches
Data Management Covers:
EDC data collection
Query resolution
Database validation
Data cleaning
At ClinAxis Research, we implement advanced data management systems to ensure accurate, audit-ready datasets.
Step 7: Safety Monitoring and Pharmacovigilance
Participant safety is the highest priority.
Safety Management Includes:
Adverse Event (AE) reporting
Serious Adverse Event (SAE) handling
Data Safety Monitoring Board (DSMB) coordination
Ongoing risk evaluation
CROs ensure real-time safety tracking and regulatory reporting within required timelines.
Step 8: Clinical Trial Phases Explained
Clinical trials typically progress through four main phases:
Phase I – Safety and Dosage
Small group of healthy volunteers
Evaluates safety, tolerability, pharmacokinetics
Phase II – Efficacy and Side Effects
Larger patient group
Assesses effectiveness and optimal dosing
Phase III – Large-Scale Confirmation
Hundreds to thousands of participants
Confirms effectiveness
Monitors adverse reactions
Compares to standard treatments
Phase IV – Post-Marketing Surveillance
Conducted after regulatory approval
Long-term safety and effectiveness monitoring
ClinAxis Research provides support across all phases of clinical development.
Step 9: Statistical Analysis and Reporting
After data collection is complete:
Activities Include:
Database lock
Statistical analysis per predefined plan
Clinical Study Report (CSR) preparation
Submission-ready documentation
Our biostatistics team ensures results are scientifically sound and regulatory compliant.
Step 10: Regulatory Submission and Approval
The final stage involves submission for marketing authorization.
Submission Dossiers May Include:
New Drug Application (NDA)
Biologics License Application (BLA)
Marketing Authorization Application (MAA)
CROs compile:
Clinical data
Safety summaries
Risk-benefit analysis
Quality documentation
Successful approval allows the treatment to reach patients.
The Role of a Clinical Research Organization (CRO)
A CRO acts as a strategic partner to sponsors by:
Managing end-to-end trial operations
Reducing development timelines
Ensuring regulatory compliance
Optimizing costs
Enhancing data integrity
Minimizing risk
By outsourcing to an experienced CRO like ClinAxis Research, sponsors gain access to specialized expertise, global networks, and operational efficiency.
Why Choose ClinAxis Research?
At ClinAxis Research, we are committed to delivering:
High-quality clinical trial management
Regulatory excellence
Patient-centric execution
Advanced data systems
Transparent communication
On-time project delivery
Our experienced team supports pharmaceutical, biotechnology, and medical device companies in navigating complex clinical development pathways.
Conclusion
Clinical trials are complex, multi-stage processes requiring careful planning, strict regulatory adherence, and expert execution. Clinical Research Organizations play a vital role in transforming innovative ideas into approved therapies that improve lives worldwide.
From protocol design to regulatory submission, ClinAxis Research ensures every step is handled with scientific rigor, operational excellence, and patient safety at the forefront.
Partner With Us
For clinical trial collaborations or inquiries:
📩 Contact us at: info@clinaxisresearch.com
🌐 Website: https://clinaxisresearch.com/
Let’s accelerate innovation together.
Contact
Reach out for clinical research support
Phone : 91+9494320540
info@clinaxisresearch.com
© 2025. All rights reserved.
